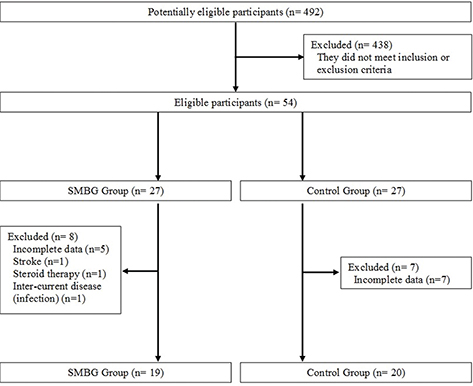
FIG 1. Flow-chart of the study design.
Original Article
Bruno Madeo1, Chiara Diazzi1,2, Antonio R.M. Granata1, Marwan El Ghoch3, Carla Greco1, Stefania Romano2, Sara Scaltriti4, and Vincenzo Rochira1
1Unit of Endocrinology, Department of Internal Medicine, Endocrinology, Metabolism, and Geriatrics, Azienda Ospedaliero-Universitaria di Modena, Ospedale Civile di Baggiovara, Modena, Italy
2Azienda USL of Modena, Modena, Italy
3Department of Nutrition and Dietetics, Faculty of Health Sciences, Beirut Arab University, Beirut, Lebanon
4Azienda USL of Reggio Emilia, Reggio Emilia, Italy
The effect of self-monitoring of blood glucose (SMBG) on glycemic control with regard to non-insulin-treated Type 2 diabetes mellitus (NIT-Type 2 DM) is still a controversial topic. Against this backdrop, we sought to compare the effect of a continuous short-term SMBG schema with as-usual treatment, based on changes in oral antidiabetic treatment in patients with poorly controlled Type 2 DM. We reviewed 492 NIT-Type 2 DM record charts, selecting 27 patients, with poor glycemic control, who were thought to self-monitor their blood glucose levels (SMBG group). We then compared them with 27 patients treated with modifying drugs or diets to achieve and maintain the glycemic target (Control Group). Haemoglobin A1c (HbA1c) and fasting plasma glucose (FPG) were evaluated at baseline, after 3 and 6 months. HbA1c values decreased after 3 and 6 months in the SMBG group (P < 0.001 on both occasions) and in the control group (P < 0.05 and P < 0.01, respectively), but without a significant difference between the two groups when compared at the same time. The FPG progressively decreased in both groups, reaching a significant difference in the SMBG group after 3 months and in the control group after 6 months, and without a significant difference between the two groups. The SMBG schema used in our study could be adopted for target groups before proceeding to the next therapeutic enhancement drug step, representing a useful tool that can help diabetic patients in raising awareness of and treating their disease.
Keywords: glycated haemoglobin, non-insulin-treatmented type 2 diabetes, self-monitoring of blood glucose
Type 2 diabetes mellitus (Type 2 DM) is an extremely complex and chronic illness which usually requires continuous medical care.1 Its prevalence has been increasing steadily all over the world.1 The reduction in risk factors and the achievement of long-term good glycemic control are the two main goals of its management and in the prevention of short- and long-term complications.1
To the best of our knowledge, significant evidence already exists and supports a range of interventions to improve diabetes outcomes.1 Further, it has been widely demonstrated that the glycemic control in Type 2 DM depends on different factors such as: (1) the disease’s natural evolution, (2) the progression of beta-cell failure, (3) the occurrence of concomitant diseases (cancers, infections, endocrine diseases, etc.), (4) the prolonged use of diabetogenic therapies (e.g., cortisone) and (5) poor patients’ compliance with treatment and inadequate lifestyle.1 In fact, good adherence to treatment remains a key strategy in avoiding or delaying the need for insulin replacement.2
The use of self-monitoring of blood glucose (SMBG) is recommended for Type 1 diabetes mellitus (Type 1 DM) and insulin-treated Type 2 DM.1 However, the role of SMBG in patients with non-insulin-treated Type 2 DM (NIT-Type 2 DM) remains controversial,1 due to conflicting results available in the literature, ranging from significant3–10 to absent11–14 improvements in haemoglobin A1c (HbA1c). That said, HbA1c reduction is generally minor,4 and SMBG alone does not seem to lower blood glucose levels; this knowledge must be factored into clinical and self-management plans.1,7
However, some of these studies involve methodological limitations (i.e., HbA1c levels at baseline are nearly within normal ranges; the absence of a standardized schema for SMBG, etc.). Moreover, they did not investigate the effectiveness of SMBG, when used alone as a therapeutic strategy in improving glycemic control, and the usefulness of the optimal frequency and timing of SMBG.
With this in view, the aim of this study was to compare the effect of the use of a continuous short-term SMBG schema for a limited period with the effect of usual treatment changes in oral antidiabetic drug (OAD) treatment and doses in patients with poorly controlled Type 2 DM. Our hypothesis is that SMBG could increase patients’ awareness of the disease, in turn potentially ensuring the best adherence to treatment.
This is an observational retrospective study.
We reviewed 492 record charts of a group of patients with NIT-Type 2 DM who were under the same clinician and had been referred to the Diabetes Unit of the “Ospedale Civile Estense” Hospital in Modena, Italy. We eventually selected 27 patients, with poor glycemic control, who were thought to self-monitor their blood glucose levels (SMBG group), and then compared them with 27 patients treated by modifying their OAD regimen (change in drug and/or dose) to achieve and maintain the glycemic target (control group) (Figure 1).

FIG 1. Flow-chart of the study design.
The patients in the SMBG group were selected from among those patients and handed a blood glucometer and a diary to collect blood glucose levels to highlight any secondary failure or low compliance according to the usual clinical practice in our unit.
In fact, these patients did not receive instructions either to interpret the SMBG data or to modify either their lifestyle habits (diet, physical activity) or the dose of the drug they were being treated with, that is, the dose was left unchanged. The patients in the SMBG group monitored their blood glucose according to the following regimen, which is arbitrarily used in the Diabetes Unit: three readings per day (one fasting control before breakfast and two postprandial controls after lunch and after dinner) over 2 days per week (one on working days and one at the weekend) (Figure 2), with the days when the readings are taken changed every week. The patients were also asked to perform an additional measurement whenever symptoms suggesting hypoglycemia or severe hyperglycemia occurred.

FIG 2. Examples of focussed SMBG regimen: a comprehensive approach is to perform three tests per day (fasting, post-lunch and post-supper), two times per week (one variable from Monday to Friday representative of a weekday and one variable from Saturday to Sunday representative of a weekend day) and additional measurement in the event of suspected hypoglycemia or severe hyperglycemia.
Clinical and biochemical data were generally collected after 3 (Visit 2) and 6 months (Visit 3).
We selected 54 record charts of subjects (25 males and 19 females) affected by Type 2 DM with poor glycemic control (HbA1c > 7%, 53 mmol/mol) and assigned patients to one of the two groups in the study according to the clinical approach used by the diabetologist (Figure 1): 27 subjects in the SMBG group and 27 in the control group. The inclusion criteria were the following: (1) diagnosis of Type 2 DM over a year ago, (2) being treated with OAD agents not reaching ceiling doses, (3) age > 45 years and (4) previous insulin administration for less than 1 week. The exclusion criteria were the following: (1) Type 1 DM, (2) secondary DM, (3) serious diabetes complications, (4) psychiatric illness, (5) history of alcohol or drugs abuse and (6) pregnancy. The adherence to glucose monitoring was evaluated by collecting data from the home diary and recorded by the blood glucometer, as reported on the record chart of selected patients. Compliance was defined as a monitoring frequency greater than 80% of the planned controls. Data from patients who did not comply with the suggested monitoring regimen were excluded from the study.
On being diagnosed with DM, all the selected subjects underwent a nutritional assessment and received a home dietary schema. Physical activity was low to moderate for both groups of subjects.
The data from 39 of the 54 record charts were considered for final evaluation (Figure 1). Fifteen record charts (n = 8 in the SMBG group and n = 7 in the control group) were excluded because of incomplete data, prolonged use of steroid therapy, major vascular events (cerebral stroke) and intercurrent disease (infection) (Figure 1).
All blood samples were collected at 8.00 a.m. after an overnight fast.
HbA1c, blood glucose, microalbuminuria, total cholesterol, high density lipoproteins cholesterol (HDL cholesterol) and triglycerides were measured by using commercially available kits.
Statistical analysis was performed by using the “Statistical Package for the Social Sciences” software for Windows (Version 16.1; SPSS Inc., Chicago, IL).
Blood glucose, HbA1c, weight and body mass index (BMI) were the analysed parameters.
Statistical approach was performed by using per protocol analysis (19 subjects in the SMBG group and 20 in the control group).
Baseline parameters between the two groups (SMBG group vs. control group) were compared by two-side Analysis of Variance (ANOVA) univariate analysis. Similarly, the parameters obtained after 3 and 6 months in each of the two groups (SMBG group and control group) were compared with baseline values by using two-side ANOVA univariate analysis. Moreover, the change (increase or decrease) from baseline was calculated for each variable considered. The endpoint change difference between two groups was evaluated by two-side ANOVA univariate.
Categorical data were compared between SMBG and control groups by using Fisher’s exact test.
For all comparisons, P values < 0.05 were considered statistically significant.
Written informed consent for participation in the study was obtained from each subject. The local institutional review board (Comitato Etico di Modena) approved the study (N. 212/16).
The analysed data concern only the 39 subjects (19 in the SMBG group and 20 in the control group) with complete data (Figure 1). The two groups did not differ in terms of the baseline parameters (Table 1).
The data for the SMBG group are from 19 patients: seven females (37%) and 12 males (63%), mean age 65 ± 7.7 years [mean ± standard deviation (SD)], mean duration of diabetes 10.6 ± 6.8 years (mean ± SD) (Table 1). The Control group data are from 20 patients: seven females (35%) and 13 males (65%), mean age 62.7 ± 9.6 years (mean ± SD), mean duration of diabetes 9.8 ± 9.1 years (mean ± SD) (Table 1).
All the patients were receiving OAD treatment, that is, a combined oral therapy for the majority of the subjects (63.2% of the SMBG group and 60.0% of the control group) (Table 1). The antidiabetic agents administrated as monotherapy were: metformin for six subjects (31.6%) from the SMBG group and six subjects (30.0%) from the control group; sulphonylurea for one subject (5.3%) from the SMBG group and two subjects (10.0%) from the control group (Table 1).
Moreover, the control group comprised a lower number of patients with diabetic retinopathy (one vs. three patients) (Table 1). Finally, subjects in the control group revealed a trend for higher triglycerides without a statistical difference, compared with the SMBG group.
In both groups, no episodes of severe hypoglycemia were reported during the period examined on the record charts.
HbA1c values progressively and significantly decreased after 3 and 6 months from baseline, both in the SMBG group [8.4 ± 0.6 vs. 7.7 ± 0.7% (68 ± 17 vs. 61 ± 16 mmol/mol), P < 0.001, and 8.4 ± 0.6 vs. 7.5 ± 0.7% (68 ± 17 vs. 58 ± 16 mmol/mol), P < 0.001, respectively] and in the control group [8.5 ± 1.0 vs. 8.0 ± 1.1% (69 ± 13 vs. 64 ± 11 mmol/mol), P < 0.05 and 8.5 ± 1.0 vs. 7.7 ± 1.0% (69 ± 13 vs. 61 ± 13 mmol/mol), P < 0.01, respectively] (Figure 3).

FIG 3. Change in HbA1c levels after 3 and 6 months in self-monitoring of blood glucose (SMBG) group and control group. The dark grey column represents HbA1c value at baseline; grey column represents HbA1c value at 3 months; and white represents HbA1c value at 6 months. Values are represented as mean ± standard deviation (SD). *ANOVA was used.
HbA1c value changes at the third and sixth months from baseline were not significantly different between the two groups (Table 2).
| Variable | SMBG group | Control group | P value for difference between groups |
|---|---|---|---|
| HbA1c | |||
| Baseline | |||
| % | 8.4 ± 0.6 | 8.5 ± 1.0 | |
| mmol/mol | 68 ± 17 | 69 ± 13 | |
| 3 months | |||
| % | 7.7 ± 0.7 | 8.0 ± 1.1 | |
| mmol/mol | 61 ± 16 | 64 ± 11 | |
| Change | −0.7 ± 0.5 | −0.5 ± 1.1 | 0.345 |
| 6 months | |||
| % | 7.5 ± 0.7 | 7.7 ± 1.0 | |
| mmol/mol | 58 ± 16 | 61 ± 13 | |
| Change | −0.9 ± 0.7) | −0.8 ± 1.1 | 0.620 |
| Glycemia (mg/dL) | |||
| Baseline | 175.0 ± 33.8 | 188.2 ± 46.1 | |
| 3 months | 163.0 ± 25.8 | 165.4 ± 38.1 | |
| Change | −12.1 ± 18.7 | −22.9 ± 54.0 | 0.865 |
| 6 months | 160.3 ± 31.4 | 159.1 ± 33.3 | |
| Change | −14.7 ± 42.6 | −29.2 ± 55.4 | 0.833 |
| BMI (kg/m 2 ) | |||
| Baseline | 28.1 ± 4.3 | 28.9 ± 4.6 | |
| 3 months | 28.0 ± 4.4 | 29.0 ± 4.6 | |
| Change | −0.1 ± 0.3 | 0.1 ± 0.4 | 0.458 |
| 6 months | 27.9 ± 4.4 | 29.1 ± 4.6 | |
| Change | −0.2 ± 0.5 | 0.1 ± 0.5 | 0.496 |
| P-value, for difference between the two groups; BMI, body mass index; SMBG, self-monitoring of blood glucose. Values are means (standard deviations). |
|||
A progressive fasting plasma glucose (FPG) decrease was observed in both groups during the study. However, this glycemic improvement was statistically significant in the SMBG group only at the third month versus baseline (175 ± 33.8 vs. 163 ± 25.8, P < 0.05), while it was only significant in the control group at the sixth month versus baseline (188.2 ± 46.1 vs. 159.1 ± 33.3, P < 0.05) with a trend for lower glycemia at the third month versus baseline (188.2 ± 46.1 vs. 165.4 ± 38.1, P = 0.06).
Glycemia value changes at the third and sixth months from baseline were not significantly different between the two groups (Table 2).
Significant changes in weight (data not shown) and BMI (Table 2) were not observed in either of the two groups.
In this observational retrospective study, we observed that the standard schema used for SMBG improved the glycemic profile in patients with poorly controlled NIT-Type 2 DM during oral hypoglycemic treatment with no ceiling dosage and without training on how to interpret their blood glucose readings.
Controversial data are available from the literature on the role of SMBG in evaluating the effect on HbA1c,5,6,9–13 while only a few studies have investigated the effect on important clinical end points instead of HbA1c, such as improvements in diabetes-related morbidity and mortality in patients with NIT-Type 2 DM.8
The reason behind these discrepancies may be due to methodological differences such as inclusion criteria and the protocol of the intervention adopted: in particular, (1) the glycemic control at baseline, (2) the duration of the disease and (3) training and support concerning the timing, interpretation and use of results to change drug prescriptions or potential corrections of patients. For instance, in the diabetes glycaemic education and monitoring study trial, poor SMBG efficacy in glycemic control could be related to an almost well-controlled glycemic profile [mean HbA1c 7.5% (58 mmol/mol)] for patients with NIT-Type 2 DM12,15 at the time of enrolment. However, to achieve significant results, a larger sample of patients is needed.10 In contrast with the present study, patients with poorer glycemic control were selected. In fact, according to previous studies, a higher level of HbA1c at baseline represents a predictive factor concerning an improvement in glycemic control by SMBG,6 as confirmed by Young et al.,14 even if this effect on HbA1c is lost in long-term follow-up.14
The ESMON 1-year trial13 showed that SMBG did not improve glycemic control in NIT-Type 2 DM, but the subjects in this trial had had a recent diagnosis of diabetes, while, in this study, patients had a longer duration of illness. These results only support the conclusion that a rapid and major improvement in glycemic control, due to the initiation or aggressive use of antidiabetic agents, could limit the contribution of SMBG to disease management.
Unlike other several studies suggesting the effectiveness of SMBG in improving glycemic control in patients with NIT-Type 2 DM,3–7,9,16 in our case, patients were not given any information about the blood glucose target to be achieved or changes in lifestyle (diet, physical activity) to be implemented. Because we did not monitor adherence to therapy, physical activity and changes to diet of patients with validated tools, we can only speculate that the improvement obtained by SMBG in our study could be the result of enhancing motivation and maintaining adherence to diet, physical activity and drug regimens, informed by an awareness of abnormal glucose values. Taking into account that many patients are unaware of the disease, because Type 2 DM is not generally accompanied by significant symptoms, as is the case with other chronic diseases. This lack of significant symptoms and the consequential lack of awareness of the disease could explain the poor adherence to therapy in most patients.2 However, it is noteworthy that all our patients were not administrated a ceiling dosage; thus, they were not involved in a secondary failure to oral hypoglycemic agents, and it could be assumed that such results would have been out of reach if our patients had been at the ceiling dosage. Moreover, another possible explanation of SMBG efficacy in relation to glycemic control in our study could be the “novelty procedure effect.” Accordingly, our patients were new users of the SMBG procedure, and this could be a short-term effect.17
A further important aspect was the SMBG schema used among our patients. Most previous studies provide information on the protocol (intensity and frequency) used5,12 or use a standard protocol, merely to indicate the total number of daily or weekly blood glucose checks.6,13 However, the usefulness, optimal frequency and timing of SMBG in patients with NIT-Type 2 DM remain unclear. In our study, we used a standard self-control regimen as previously described (Figure 2), which, although arbitrary, was based on a criterion of maximum reading representation, with minimal discomfort for the patient. Considering that SMBG can raise the level of stress,18 we have reduced the frequency of measurements to maintain the highest representation of glycemic variability linked to different daily activities and provide a good representation of the blood glucose variability during the week under ordinary conditions, with the least possible number of measurements. Hence, due to the improvement in glycemic control achieved with the proposed SMBG schema, we suggest its adoption as a strategy in patients with poorly controlled NIT-Type 2 DM, before implementing the drug therapy.
Furthermore, as reported by other studies,5 in our case, body weight was not reduced during the follow-up. This could indirectly indicate that improvements in blood glucose profiles seen in our study were not because of weight loss, as the latter is considered to be an effective therapeutic strategy in the treatment of Type 2 DM.1
Our study involves some limitations, the foremost of which are the retrospective design, the short-term duration of follow-up and the small sample size. Accordingly, even though the patients included in the study were to minimize selection bias, the latter may be not completely avoided due to the retrospective nature of the study. Moreover, data were obtained from a single unit by applying one treatment protocol, which meant that external validation was required. Hence, future research should be designed by taking these limitations into account. Nevertheless, important clinical implications can be evinced from the study. First, taking into account the most recent literature suggesting that self-monitoring may be useful when coupled with a treatment adjustment programme,1 clinicians should be aware of the effectiveness of SMBG as a strategy that can improve glycemic control in patients with poorly controlled Type 2 DM receiving OAD agents with no ceiling dosage. In addition, this finding should be emphasized and discussed with patients as a possible motivational aid.
In conclusion, the standard SMBG regimen used in our study could be an option to treat target groups, in particular, patients with poorly controlled NIT-Type 2 DM (HbA1c > 8%) and not with a recent diagnosis, before proceeding to the next therapeutic enhancement drug step. This is because SMBG could be a useful tool, which can help diabetic patients in raising awareness of and treating their disease.
This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
We thank Dr Daniele Santi for his precious support on statistical analysis.
The datasets supporting the findings of this study are available from the corresponding author [B.M.] on request.
The study was carried in compliance with ethical standards.
The authors declare that they have no conflict of interest.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
The local Institutional Review Board (Comitato Etico di Modena) approved the study (N. 212/16).
For this type of study formal consent is not required; however, written informed consent for participation in the study was obtained from each subject.